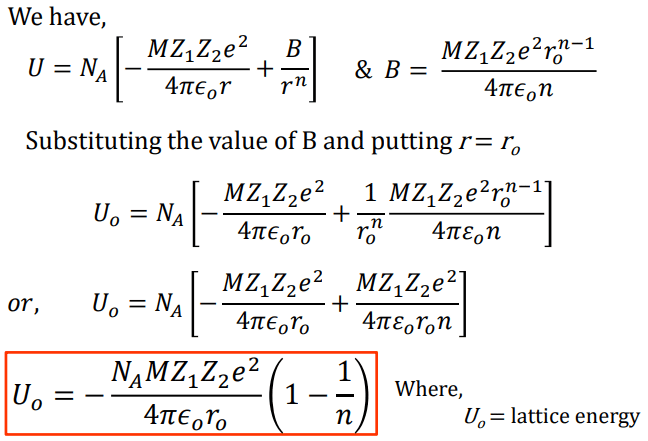 
The teams’ findings emphasize the need to consider negative-energy states when performing high-precision studies of the interactions between light and matter. Sergey Porsev at the University of Delaware and his colleagues explain this feature mathematically Li-Yan Tang and colleagues at the Chinese Academy of Science calculate the contributions both for strontium atomic clocks and for magnesium, calcium, and cadmium ones. These contributions greatly impact the atoms’ magnetic-dipole polarizability but not their electric-quadrupole polarizability. The two teams resolve this discrepancy by considering previously ignored contributions to the polarizabilities from so-called negative-energy states of the atoms, which are predicted by quantum field theory. But until now, the predicted values of these polarizabilities have conflicted with those measured in experiments. The size of this shift depends on two properties of the atoms, known as their magnetic-dipole and electric-quadrupole polarizabilities. The interaction between the trapped atoms and the lattice’s electromagnetic field induces a shift in the energies of the atomic energy levels used for timekeeping. In the most precise strontium atomic clocks, a lattice of intersecting laser beams, all with the same “magic” wavelength, traps strontium atoms in certain locations. The results could facilitate the use of strontium timekeepers in the space-based detection of gravitational waves, for example. This disparity has now been rectified by two independent research teams. But the precision of these clocks has been limited by an enduring inconsistency between predictions of their properties and corresponding observations. The value of the Madelung constant has been calculated for all common crystal structures by summing the contributions of all the ions in the crystal lattice.Strontium atomic clocks have important applications, ranging from tests of general relativity to searches for possible variations in fundamental constants. The lattice energy of the ionic crystal is inversely proportional to the Inter-ionic distance and directly proportional to the product of charges of the ions, Madelung constant, and Born exponent. Where N A is the Avogadro constant, the number of molecules in a mole has the value 6.023×10 23 mol -1 A is the Madelung constant, which depends on the geometry of the crystal. Total energy = Attractive energy + Repulsive energyįor one mole of the ionic crystal U = E total N A This is the Born Lande equation. 
M = Madelung constant, which is related to the geometry of the crystal Repulsive force where, B = constant, A portion of three-dimensional cubic lattice and its unit cell Where Z + and Z – are the charges on the positive and negative ions,Īttractive energy for a simple lattice of the crystal 
The ions are treated as point charges, and the electrostatic energy E between two ions of opposite charge is calculated. Theoretical values for lattice energy may be calculated. Lattice energies cannot be measured directly, but experimental values are obtained from thermodynamics data using the Born Haber cycle. Lattice energy is defined as the energy released in the process when the constituent ions are placed in their respective positions in the crystal lattice or, the amount of energy required to separate the solid ionic crystal into its constituent ions. Schematic representation of lattice energy at inter-ionic distance r o For sodium chloride, the lattice energy, U, is equal to the enthalpy change for the reaction. The lattice energy (U) of a crystal is the energy that evolved when one gram of the crystal is formed from gaseous ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
